Interested in this topic? By joining CR4 you can "subscribe" to
this discussion and receive notification when new comments are added.
Good Answers:
|
| Previous in Forum: what is the LPG Vapour calculation formula | Next in Forum: Weld ability between A182 F51 and A182 F316 |
Advertisement
Got Something to Say? |
||
Search this Forum |
||
|
Chemical & Material Science: |
||
Site Directories |
||
|
Forum Directory Blog Directory User Group Directory RSS Feeds CR4 Rules of Conduct CR4 FAQ Site Glossary Who's Online (102 right now) Show Members |
||
Advertisement


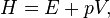


Good Answers:
"Almost" Good Answers: